Vol-1 Issue-1 Year-2026
Dr. Dr C. Sharath Babu1*, Dr. Bhavya Sri Kakumanu2, Dr. Naveen K3, Dr. Shrushti S Nagar4*, Dr R Viswachandra5
¹ MDS, FICOI, Prosthodontist and Implantologist, Head of Department, Dental Wing, Medicover Hospitals, Hi-tech City, Hyderabad, India
² BDS, General Dentist, Medicover Hospitals, Hi-tech City, Hyderabad, India
³ MDS, Oral and Maxillofacial Surgery, Medicover Hospitals, Hi-tech City, Hyderabad, India
⁴ BDS, Postgraduate in Periodontology and Implantology, SVS Institute of Dental Sciences, Mahbubnagar, India
⁵ MDS, DNB, Periodontics and Implantology, Professor and Head of Department, SVS Institute of Dental Sciences, Mahbubnagar, India
Author of Correspondence: Dr. Shrushti S Nagar, Postgraduate in Periodontology and Implantology, SVS Institute of Dental Sciences, Mahbubnagar
Email: shrushti.nagar@gmail.com
KEYWORDS:
papillon lefèvre syndrome, palmoplantar keratoderma, aggressive periodontitis, multidisciplinary strategy, oral rehabilitation, digital dentistry, selective laser melting (SLM), customized subperiosteal implants, patient specific implants-PSI.
Received date-02-03-2026
Accepted date-17-03-2026
Published date -22-03-2026
Citation format-Babu CS, Kakumanu BS, Naveen K, Nagar SS, Viswachandra R. Rehabilitation of a subject with Papillon Lefèvre syndrome with patient-specific implants. J Dent Innov Med Sci. 2026;1(1):26–32.
Abstract
Background: Oral Rehabilitation of patients with severe/advanced bone loss using state-of-the-art technology i.e., Patient-Specific Implants.
In this case report we are describing the oral rehabilitation of a 19-year-old male Papillon Lefèvre Syndrome (PLS) patient, who had insufficient remaining bone with Malo prosthesis supported by Patient Specific Implants (PSI) in both Maxilla and Mandible which is first of its kind. The patient’s functional, esthetic and psychological health all improved greatly as a result of prosthesis.
Conclusion: Use of PSI in maxillofacial surgery has consistent outcomes enabling more accurate reconstruction of maxillofacial defects and the complications that are usually encountered with non custom-made implants are not observed.
Introduction
The Papillon lefèvre syndrome was originally described by 2 French physicians, Papillon and Lefevre in 1924. This syndrome is inherited as an autosomal recessive trait with a prevalence of 1 to 4 cases per million. This disorder is marked by palmoplantar hyperkeratosis and severe early-onset periodontitis, leading to the premature loss of both primary and permanent teeth. In the present case, the dermatological and the dental findings along with the history strongly suggest it to be the Papillon lefèvre syndrome. The differential diagnosis includes acrodynia (pinks disease), hypophosphatasia, cyclic neutropenia, keratosis punctata, Howel – Evanssyndrome, Grither syndrome. It differs from acrodynia with the absence of erythrocyanosis, muscle pain, tachycardia, psychic disturbances and teeth erupting prematurely with dystrophic enamel. Hypophosphatasia is differentiated from this syndrome by the presence of bowing of femur and tibia, knock – knee, enlarged wrists, hypoplastic teeth and increased amounts of phosphoethanolamine in the urine. Unlike PLS, Howe-Evans syndrome, Greither’s syndrome, keratosis punctata do not have periodontopathic; cyclic neutropenia does not present with palmoplantar hyperkeratosis.[3] Treatment of PLS patients is symptomatic and often ineffective. Early management of the periodontal destruction includes eliminating the reservoir of causative organisms, using conventional periodontal treatment, oral hygiene instructions, antiseptic mouth rinses and systemic antibiotic therapy. During the growth period, prosthetic therapy is necessary, and periodontal maintenance may allow the patient to maintain a part of their dentition. Failure to respond to antibiotic therapy is significant in the treatment of PLS. The prosthetic approach is an age specific treatment. At an early age, during deciduous or mixed dentition period extraction of all the primary and permanent teeth followed by replacement with removable prosthesis. Some authors described the use of titanium implants [7]. Bilal Ahmed et al described the treatment plan of extracting the remaining teeth and giving a removable complete denture with bilateral balanced occlusion in a 17-year-old female patient [8]. Leila Ahmadian et al used premade endosseous dental implants in a 21-year-old female patient [9]. For partially or fully edentulous patients, endosseous dental implants offer a reliable prosthetic solution, but they require adequate bone height and width for successful placement. In cases of severe bone atrophy, regenerative procedures like bone grafting, guided bone regeneration, ridge splitting, distraction osteogenesis, and sinus augmentation are often necessary. However, these complex techniques are costly, time-consuming, and may still result in residual facial asymmetry due to limitations in addressing specific defects. Digital revolution has transformed the dental field significantly in the current day. New acquisition techniques, new materials, processing and fabrication tools like computer-assisted design/computer assisted manufacturing (CAD/CAM) software have significantly changed the field of dentistry. In the case of 3D printers, unlike resin materials that were available in the early days, it is currently possible to print t itanium materials that have already been verified for biocompatibility as dental implants. Today’s innovative Additive Manufacturing techniques such as direct metal laser sintering (DMLS) and selective laser sintering (SLS) and selective laser melting (SLM) processes make it possible to create customized grids and implants that precisely match each patient’s unique anatomical needs. By using these technologies, some of the older methods, such as subperiosteal implants, can be revisited and reinterpreted in a contemporary and digital manner [10]. By utilizing the modern technologies, combining the traditional subperiosteal implant designs with 3D imaging and printing can save treatment time and support fixed prostheses in advanced / severe bone loss / atrophy cases. Since they are customized for each patient, they restore the defect in a way that ensures both functional and aesthetic satisfaction and well-being.
Conclusion
The prosthetic rehabilitation to achieve the desired anatomical, functional and esthetic requirements still remains a challenge when there is insufficient bone left. The traditional approaches are unable to provide complete justification. Each of these needs can be addressed by combining the most recent developments in digital dentistry with conventional treatment methods. PSI is one such treatment modality. The use of PSI for maxillofacial reconstruction has great patient satisfaction, predictable results, and no usual complications that come with non-custom-made implants. The biggest disadvantage being its expensive cost.
Conflict of Interest: Nil
Ethical consent was obtained from the patient. Institutional Review Board approval was not required for this study.
- Wani AA, Devkar N, Patole MS, Shouche YS. Description of two new cathepsin C gene mutations in patients with Papillon-Lefèvre syndrome. J Periodontol. 2006; 77:233-7.
- Hart TC, Shapira L. Papillon-Lefèvre syndrome. Periodontol 2000. 1994; 6:88-100.
- Sreeramulu B, Shyam ND, Ajay P, Suman P. Papillon-Lefèvre syndrome: clinical presentation and management options. Clin Cosmet Investig Dent. 2015 15; 7:75-81.
- Pareek SS, Al-Aska AK. Papillon-Lefevre syndrome. A report of six cases in one family. Int J Dermatol. 1986; 25:638-41.
- Phull T, Jyoti D, Malhotra R, Nayak S, Modi H, Singla I, P A. Diagnosis and Management of Papillon-Lefevre Syndrome: A Rare Case Report and a Brief Review of Literature. Cureus. 2023 11;15: e43335.
- Joshi S, Salema HJ, Pawar S, Nair VS, Koranne V, Sane VD. Patient-Specific Implants in Maxillofacial Reconstruction – A Case Report. Ann Maxillofac Surg. 2023; 13:258-261.
- Abdulwasi H, Dhanrajani PJ, Jiffry A. Papillon-Lefèvre syndrome. Reappraisal of etiology, clinical features and treatment. II. Oral rehabilitation using osseointegrated implants. Indian J Dent Res. 1996; 7:63-70.
- Ahmed B. Prosthodontic rehabilitation of Papillon Lefevre Syndrome. J Coll Physicians Surg Pak. 2014; 24 Suppl 2: S132-4.
- Ahmadian L, Monzavi A, Arbabi R, Hashemi HM. Full-mouth rehabilitation of an edentulous patient with Papillon-Lefèvre syndrome using dental implants: a clinical report. J Prosthodont. 2011; 20:643-8.
- Javaid M, Haleem A. Current status and applications of additive manufacturing in dentistry: A literature-based review. J Oral Biol Craniofac Res. 2019; 9:179-185.
Figures
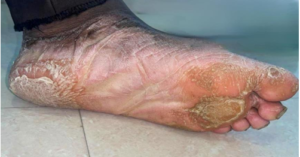
Figure 1a: Extraoral image – Left foot dorsal surface
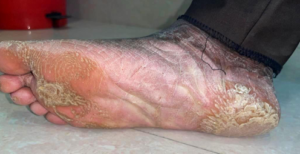
Figure 1b: Extra oral image – Right foot dorsal surface
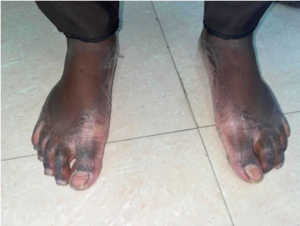
Figure 1c: Extra oral image – Both feet
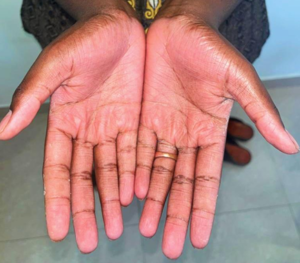
Figure 1d: Extraoral image – Palms
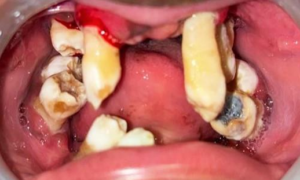
Figure 2a: Preoperative Intra-oral image
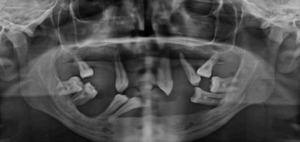
Figure 2b: Preoperative OPG
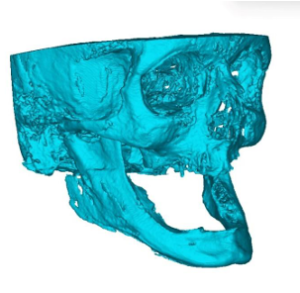
Figure 3a: 3D resin model made from DICOM data from CT
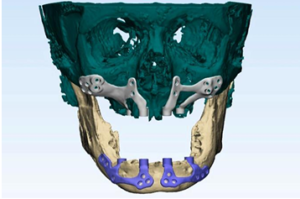
Figure 3b: Customized subperiosteal implants designed virtually 3D virtual bone model
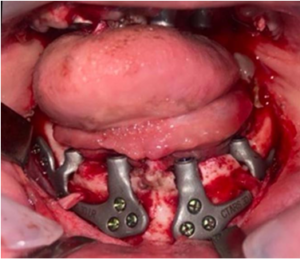
Figure 4: Intraoperative image
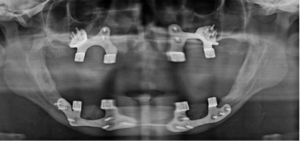
Figure 5: Post-operative OPG after placement of implants
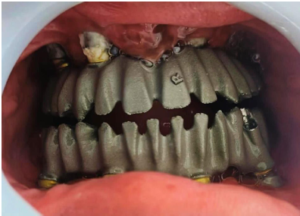
Figure 6: Metal trail
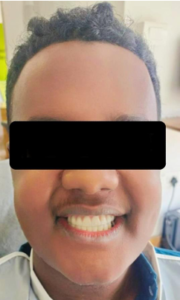
Figure 7a: Smile after final prosthesis placement
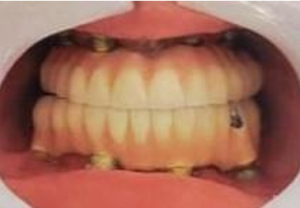
Figure 7b: Intraoral image after final prosthesis placement
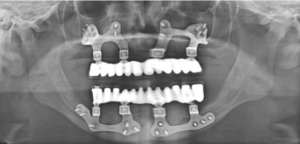
Figure 8: Postoperative OPG after final prosthesis placement


